Most people don’t realize that a small radioactive source sits quietly in their hallway, emitting alpha particles 37,000 times per second. It’s been there for years, possibly decades, and it’s one of the most successful life-saving devices ever invented. The humble smoke detector contains about 0.3 micrograms of americium-241—a byproduct of nuclear reactors—and understanding how it works reveals a fascinating intersection of nuclear physics, electrical engineering, and fire science.
The Accidental Discovery
In the late 1930s, Swiss physicist Walter Jaeger was attempting to build a sensor for poison gas. His approach used an ionization chamber: air molecules between two charged plates would be ionized by a radiation source, creating a small electrical current. When poison gas entered, he expected it to bind to the ions and change the current.
The device failed to detect gas at low concentrations. Frustrated, Jaeger lit a cigarette. The meter on his instrument suddenly registered a drop in current. The smoke particles from his cigarette had done what poison gas could not—they attached to the ionized air molecules and disrupted the current flow. Jaeger had accidentally invented the ionization smoke detector.
This serendipitous discovery would take decades to reach homes. The first commercial ionization detectors appeared in 1951, but they were large, expensive, and limited to industrial facilities. It wasn’t until 1965 that Duane D. Pearsall and Stanley Bennett Peterson developed the first affordable, battery-powered unit suitable for home use. By 1975, mass production had begun.
How Ionization Actually Works
The heart of an ionization smoke detector is a dual-chamber system containing a tiny amount of americium-241 oxide—typically about 37 kilobecquerels (37,000 decays per second), equivalent to roughly 0.3 micrograms of the isotope. Am-241 was chosen for specific reasons: it’s an alpha emitter with a half-life of 432.6 years, meaning it remains active for the device’s lifetime, and its alpha particles have enough energy to ionize air but can’t penetrate human skin.

Image source: Wikipedia - Smoke Detector
When an americium-241 nucleus decays, it ejects an alpha particle—a helium nucleus consisting of two protons and two neutrons—with an energy of about 5.4 MeV. This particle collides with air molecules in the chamber, primarily nitrogen and oxygen, stripping electrons and creating ion pairs. The process looks like this:
$$\text{N}_2 + \alpha \rightarrow \text{N}_2^+ + e^- + \alpha'$$The alpha particle continues along a short path (typically 3-5 cm in air), creating approximately 30,000 ion pairs before it exhausts its energy and becomes a harmless helium atom.
Two metal plates in the chamber sit at different voltages. The positively charged ions drift toward the negative plate, while electrons move toward the positive plate. This ion flow creates a continuous electrical current—typically on the order of picoamperes. A buffer amplifier with extremely low input bias current (often less than 1 pA) measures this tiny current.
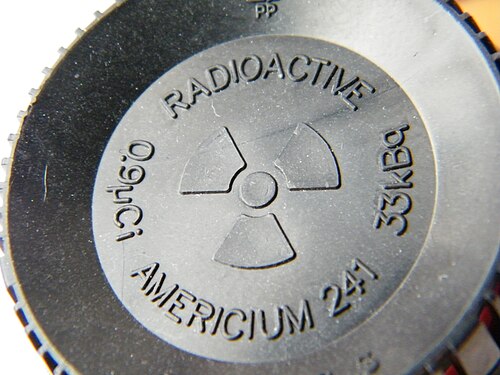
Image source: Wikipedia - Americium-241
The critical innovation is the dual-chamber design. One chamber is open to ambient air, while a reference chamber is partially sealed. Both chambers receive the same radiation and are affected equally by temperature, humidity, and the gradual decay of the americium source. The collector plate between them measures the difference in current between the two chambers.
When smoke enters the open chamber, smoke particles—much larger than ionized air molecules—attach to the ions. These combined particles have greater mass but the same thermal energy, so they move more slowly. The reduced mobility shows up as a drop in current. The reference chamber, protected from direct airflow, maintains its baseline current. The resulting imbalance triggers the alarm.
Why Alpha Radiation Is Safe Inside Your Home
The safety of ionization detectors comes down to the nature of alpha particles. At 5.4 MeV, an alpha particle from americium-241 can be stopped by a sheet of paper, a few centimeters of air, or the outer layer of human skin. The plastic housing of a smoke detector provides complete shielding.
The radiation dose from a typical ionization detector is estimated at 9-50 nanosieverts per year. For comparison, eating a banana exposes you to about 100 nSv (from potassium-40), and the average person receives about 3.6 millisieverts annually from natural and manmade sources combined. The detector’s contribution is genuinely negligible.
The real risk would come from inhaling or ingesting the americium source—hence the warnings against dismantling or burning smoke detectors. The sealed foil source is designed to remain intact even in a house fire.
The Photoelectric Alternative
Not all smoke detectors use nuclear physics. Photoelectric detectors, invented in 1972 by Donald Steele and Robert Emmark, rely on an entirely different principle: light scattering.

Image source: Wikipedia - Smoke Detector
Inside a photoelectric detector, an LED (typically infrared at 850 nm) emits light into a sensing chamber. The chamber geometry is designed so that the light beam passes through the chamber without hitting the photodetector—a photodiode positioned at an angle to the light path. This is the dark chamber state.
When smoke particles enter, they scatter light in all directions. Some of this scattered light reaches the photodetector, which generates an electrical current proportional to the light intensity. The phenomenon is known as the Tyndall effect—the same reason you can see a laser beam through fog or dusty air.
The scattering efficiency depends on the relationship between particle size and light wavelength. According to Mie scattering theory, particles with diameters comparable to or larger than the wavelength scatter light most effectively. For an 850 nm infrared LED, particles in the 0.5-10 micron range are ideal.
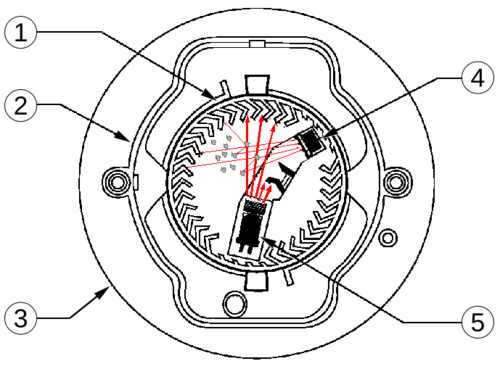
Image source: Wikipedia - Smoke Detector
Why Fire Type Matters
The reason both technologies exist—and why experts recommend using both—comes down to smoke particle physics. Different fire stages produce different particle sizes.
Flaming fires burn hot and fast, producing tiny particles between 0.01 and 0.4 microns. These particles are too small to scatter light efficiently, but they’re perfect for disrupting ion flow. Ionization detectors respond 30-60 seconds faster to flaming fires than photoelectric models.
Smoldering fires burn cooler and slower, producing larger particles between 0.4 and 10 microns—exactly the sweet spot for light scattering. Photoelectric detectors can respond 47-53 minutes faster to smoldering fires than ionization models. In a smoldering fire, those extra minutes are often the difference between escape and tragedy.
The NIST study “Performance of Home Smoke Alarms” (Technical Note 1455, 2004) confirmed these differences through extensive testing. The study found that ionization alarms provided “somewhat better response to flaming fires” while photoelectric alarms provided “considerably faster response to smoldering fires.”
Where Americium-241 Comes From
The americium in smoke detectors has a nuclear origin. Inside a nuclear reactor, uranium-238 captures neutrons to form uranium-239, which beta-decays to neptunium-239, then to plutonium-239. Some plutonium-239 atoms capture additional neutrons, becoming plutonium-240 and then plutonium-241. Pu-241 beta-decays with a half-life of about 14 years to become americium-241.
The production is slow—plutonium must sit for years while Pu-241 decays into americium. The americium is then chemically separated from spent fuel or aged plutonium stocks. This connection to nuclear fuel cycles is why large-scale americium production is limited to nuclear-capable nations.
Interestingly, the same Am-241 used in smoke detectors is now being investigated for radioisotope thermoelectric generators (RTGs) for spacecraft. Its 432-year half-life makes it attractive for long-duration missions, though its lower power density compared to plutonium-238 presents challenges.
Effectiveness By The Numbers
The statistics on smoke detector effectiveness are striking. According to the National Fire Protection Association, the risk of dying in a residential fire is cut in half in homes with working smoke detectors: 0.53 deaths per 100 fires with detectors versus 1.18 deaths per 100 fires without.
Three out of five home fire deaths occur in properties without working smoke alarms—either no alarms at all (40%) or alarms that failed to operate (17%). The most common cause of failure: missing or disconnected batteries, often removed due to nuisance alarms.
This last point is significant. Ionization detectors are more prone to false alarms from cooking, which leads some occupants to disable them. Photoelectric detectors have fewer cooking-related false alarms but cost more. Some jurisdictions, including parts of Australia and several U.S. states, have moved toward requiring photoelectric detectors or dual-sensor alarms for this reason.
Modern Developments
The latest smoke detector technology combines multiple sensing methods. Modern units may include both ionization and photoelectric sensors, along with carbon monoxide detection and advanced algorithms to distinguish between real fires and nuisance sources.
Texas Instruments’ LPV801 nanopower amplifier, designed specifically for smoke detector applications, exemplifies the engineering constraints: less than 1 pA input bias current, quiescent current under 500 nA, and operation across the 0°C to 49°C range specified by UL 217. The device must run continuously for years on a 9-volt battery while detecting picoampere-level current changes.
Analog Devices’ ADPD188BI takes a different approach, using dual-wavelength photoelectric detection (470 nm blue and 850 nm infrared) to better characterize particle types. Different smoke sources scatter blue and infrared light differently, allowing more sophisticated fire-versus-nuisance discrimination.
The Bottom Line
The smoke detector on your ceiling represents one of the most successful applications of nuclear physics to everyday life. A fraction of a microgram of reactor-produced americium, decaying at 37,000 emissions per second, creates an ionization current that has saved countless lives. The photoelectric alternative, born from optics rather than nuclear science, complements it perfectly.
Neither technology is superior—each excels at detecting different fire types. The best protection comes from using both, either as separate units or combined in a dual-sensor alarm. The small radioactive source in your hallway isn’t something to fear. It’s a silent guardian that’s been watching over homes for over fifty years, one alpha particle at a time.
References
- Eason, E. (2011). “Americium Smoke Detectors.” Stanford University, PH241.
- National Fire Protection Association. “Smoke Alarms in U.S. Home Fires.” NFPA Research.
- Bukowski, R.W. et al. (2004). “Performance of Home Smoke Alarms: Analysis of the Response of Several Available Technologies in Residential Fire Settings.” NIST Technical Note 1455.
- Texas Instruments. (2016). “LPV801 Micropower Ionization Smoke Detector.” Application Report SNOA960.
- Analog Devices. (2020). “A Modern Solution to Evolving Smoke Safety Regulations.”
- U.S. Environmental Protection Agency. “Americium in Ionization Smoke Detectors.”
- World Nuclear Association. “Radioisotopes in Consumer Products.”
- U.S. Nuclear Regulatory Commission. “Ionization Chamber Smoke Detectors Containing Am-241.” NUREG/CR-1156.