On December 2, 1942, beneath the stands of a squash court at the University of Chicago, Enrico Fermi and his team achieved something humanity had never accomplished: a controlled, self-sustaining nuclear chain reaction. Chicago Pile-1, the world’s first nuclear reactor, produced just half a watt of power—barely enough to light a small bulb. Yet it demonstrated a principle that now generates about 9% of the world’s electricity, powering hundreds of millions of homes with the energy locked inside atomic nuclei.
Most people understand that nuclear power plants “split atoms” to generate electricity. Far fewer understand why you cannot simply turn off a nuclear reactor the way you flip off a light switch. This fundamental constraint—born from the physics of radioactive decay—shapes every aspect of reactor design, operation, and safety. Understanding it reveals why nuclear engineering is among humanity’s most demanding technical endeavors.
The Energy in an Atom
The power of nuclear fission comes from a counterintuitive fact: the total mass of a nucleus is less than the sum of its constituent protons and neutrons. This “mass defect” represents binding energy—the energy required to hold the nucleus together. Einstein’s equation $E = mc^2$ quantifies this relationship: a small amount of mass corresponds to an enormous amount of energy.
When a uranium-235 nucleus absorbs a neutron, it becomes unstable and splits into two smaller nuclei called fission products. This split releases approximately 200 million electron volts (MeV) of energy—roughly 50 million times more energy per atom than burning carbon in fossil fuels. The energy manifests as kinetic energy of the fission fragments (about 85%), gamma radiation, and the kinetic energy of released neutrons.
But fission releases more than energy. Each fission event typically releases 2-3 neutrons, with an average of about 2.5. These neutrons can strike other uranium nuclei, causing additional fissions. This is the chain reaction: one fission leads to more, which leads to more still.
flowchart TD
A[U-235 Nucleus<br/>+ Neutron] --> B[Unstable Compound Nucleus]
B --> C[Fission Event]
C --> D[Fission Product 1<br/>e.g., Kr-92]
C --> E[Fission Product 2<br/>e.g., Ba-141]
C --> F[2-3 Neutrons<br/>+ ~200 MeV Energy]
F --> G[Chain Reaction]
G --> A
style A fill:#ffcccc
style C fill:#ffffcc
style F fill:#ccffcc
The multiplication factor, denoted k, determines whether the reaction grows, shrinks, or stays constant. When k equals 1, the reactor is critical—the number of fissions remains steady, and power output is stable. When k exceeds 1, the reactor is supercritical, and power increases. When k falls below 1, the reactor is subcritical, and power decreases. Control systems adjust k by inserting or withdrawing neutron-absorbing control rods.
The Moderation Problem
Not all neutrons are equally likely to cause fission. Neutrons born from fission travel at about 20,000 kilometers per second—roughly 7% of the speed of light. At these energies, they are “fast” neutrons, and their probability of causing fission in U-235 is relatively low.
However, if neutrons slow down to thermal energies—about 2 kilometers per second, comparable to the thermal motion of atoms—their fission cross-section for U-235 increases dramatically. This is why most commercial reactors include a moderator: a material that slows neutrons through elastic collisions.
Water is the most common moderator because its hydrogen atoms have roughly the same mass as neutrons, making collisions efficient. Heavy water (deuterium oxide) is even more effective because it absorbs fewer neutrons, allowing reactors to run on natural uranium without enrichment. Graphite also works—the first reactor at Chicago used graphite blocks.
The choice of moderator creates a fundamental design split. Light water reactors (LWRs) need enriched uranium because ordinary water absorbs too many neutrons to sustain a chain reaction with natural uranium. Heavy water reactors like Canada’s CANDU can use natural uranium but require expensive heavy water. Each approach involves trade-offs between fuel enrichment costs and moderator costs.
The Two Dominant Designs
About 85% of the world’s nuclear electricity comes from two reactor types that emerged from 1950s submarine propulsion programs: the pressurized water reactor (PWR) and the boiling water reactor (BWR).
Pressurized Water Reactor
The PWR operates with two separate water circuits. In the primary circuit, water circulates through the reactor core at about 325°C under 150 atmospheres of pressure—high enough pressure to keep the water liquid despite temperatures well above water’s normal boiling point. This superheated, pressurized water carries heat from the reactor to a steam generator, where it transfers heat to a secondary water circuit. The secondary water boils to produce steam that drives turbines.
This separation means the turbine and condenser never contact radioactive water from the primary circuit. However, it requires enormous pressure vessels capable of containing the primary coolant, and the design introduces thermal efficiency penalties from the intermediate heat exchange.
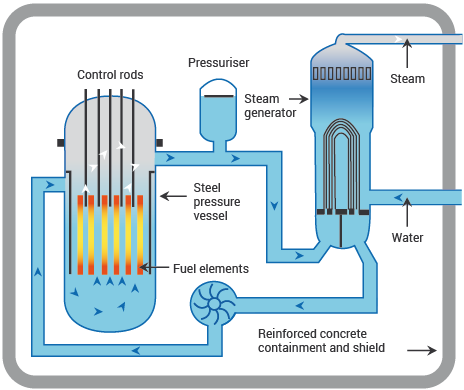
Image source: World Nuclear Association - Nuclear Power Reactors
Boiling Water Reactor
The BWR simplifies this arrangement by allowing water to boil directly in the reactor core. Steam rises through the core, passes through steam separators and driers, and flows directly to the turbine. This eliminates the expensive steam generator and secondary loop but means the turbine becomes slightly radioactive from contact with reactor water.
Most radioactivity in BWR coolant comes from nitrogen-16, formed when oxygen in water absorbs neutrons. N-16 has a half-life of only 7 seconds, so turbine rooms become safe to enter shortly after reactor shutdown.
Both designs achieve thermal efficiencies of 33-37%—meaning about two-thirds of the nuclear heat is rejected to the environment through cooling towers or bodies of water. This efficiency limit stems from fundamental thermodynamics: the Rankine cycle’s efficiency depends on the temperature difference between the heat source and heat sink.
Why Control Rods Are Not Enough
Here lies the critical insight that distinguishes nuclear from all other forms of power generation: stopping the chain reaction does not stop heat production.
When control rods absorb neutrons and halt the chain reaction, fission stops. But the fission products—the fragmented nuclei created by splitting uranium—remain intensely radioactive. These radioactive decays release energy as heat. This “decay heat” cannot be stopped by any control mechanism because it is an inherent property of the fission products themselves.
Immediately after shutdown, decay heat equals about 6-7% of the reactor’s full power output. In a typical 3,000 megawatt-thermal reactor, that means 180-210 megawatts of heat continue to be generated even with all control rods fully inserted. This drops to about 1% after a day and continues declining, but even after a year, spent fuel generates roughly 1 kilowatt per tonne.
Image source: What Is Nuclear - Decay Heat
This physics explains why Fukushima was so catastrophic. The earthquake triggered an automatic shutdown—control rods inserted successfully, and the chain reaction stopped. But the tsunami flooded the emergency diesel generators that powered cooling pumps. Without cooling, decay heat accumulated until fuel melted, hydrogen generated from zirconium-water reactions accumulated, and explosions breached containment.
timeline
title Fukushima Accident Timeline (2011)
section March 11
Earthquake strikes : Reactor scrams successfully
Tsunami arrives : Floods emergency generators
Station blackout : Loss of all cooling power
section Hours later
Core damage begins : Decay heat accumulates
Hydrogen buildup : Zirconium-steam reaction
section Days following
Hydrogen explosions : Containment breached
Radioactive release : Evacuation ordered
This is also why modern reactors incorporate multiple redundant cooling systems: battery-backed systems, steam-driven pumps that require no electricity, and in some advanced designs, natural circulation that removes decay heat through convection alone.
The Role of Delayed Neutrons
The chain reaction’s controllability depends on another subtle physics phenomenon. About 99.3% of neutrons from fission are “prompt”—released essentially instantaneously when the nucleus splits. But 0.7% are “delayed,” emitted by fission products seconds to minutes after the original fission event as they undergo radioactive decay.
This tiny fraction of delayed neutrons is crucial. If all neutrons were prompt, any deviation from exact criticality would cause power to change exponentially in milliseconds—far too fast for any mechanical control system to respond. Delayed neutrons add a slow component to the chain reaction, stretching the reactor’s response time to seconds or minutes, enabling human operators and automatic systems to maintain control.
Reactor operators never allow the reactor to become critical on prompt neutrons alone. The margin between criticality with delayed neutrons included and prompt criticality is carefully monitored and protected by automatic shutdown systems.
Safety Through Physics
Modern reactor designs incorporate inherent safety features that rely on physics rather than active systems. Light water reactors have negative temperature coefficients: as water heats and expands (or boils), its density decreases, reducing moderation and slowing the chain reaction. This negative feedback provides automatic power regulation—if temperature rises, reactivity decreases.
The disaster at Chernobyl occurred partly because the RBMK reactor design had a positive void coefficient under certain operating conditions. When coolant boiled, neutron absorption decreased and reactivity increased—exactly the wrong direction for safe operation. Combined with control rod design flaws and operator actions that disabled safety systems, this positive feedback led to a power excursion that destroyed the reactor.
Modern designs eliminated such vulnerabilities. Today’s reactors are designed so that any departure from normal operation inherently tends to return the system to a safe state, even without operator intervention.
What the Fuel Becomes
A typical fuel assembly spends three to six years in a reactor core. During this time, about 4-5% of the uranium atoms undergo fission. Some U-238 captures neutrons and transforms through beta decay into plutonium-239, which also fissions and contributes roughly one-third of the reactor’s energy output.
When fuel is removed, it contains a complex mixture: remaining uranium, plutonium, and hundreds of different fission products with varying half-lives. Iodine-131, with an 8-day half-life, produces intense radioactivity initially. Cesium-137 and strontium-90, with 30-year half-lives, dominate radioactivity after decades. Plutonium-239 has a half-life of 24,000 years.
This inventory of long-lived radioactivity creates the central challenge of nuclear waste management. Current approaches involve storing spent fuel in pools for several years (where decay heat drops substantially), then transferring to dry casks for interim storage. Long-term plans involve geological repositories—deep underground facilities where engineered barriers and stable geology isolate waste for the hundreds of thousands of years required for radioactivity to decline to safe levels.
The Scale of Operation
The numbers involved in nuclear power are staggering. A single PWR fuel assembly weighs about 655 kilograms and holds 460 kilograms of uranium. A large reactor core contains 150-250 such assemblies—over 18 million individual fuel pellets, each about the size of a pencil eraser, each precisely manufactured to within micrometers of specification.
Steam generators weigh up to 800 tonnes and contain thousands of tubes that must remain leak-tight for decades while exposed to high-pressure, high-temperature, slightly radioactive water. Reactor pressure vessels are forged from single ingots of special steel, designed to contain 150 atmospheres of pressure at temperatures above 300°C for 60 years or more.
Globally, 416 nuclear reactors operate in 31 countries, providing 376 gigawatts of electrical capacity. The United States operates 94 reactors; France generates about 70% of its electricity from nuclear. Each reactor represents an investment of billions of dollars and a commitment to decades of careful operation and oversight.
The Unavoidable Constraint
Nuclear power’s unique characteristic—the inability to simply switch it off—remains its defining challenge. This constraint drives the engineering complexity, the multiple redundant safety systems, the extensive regulatory oversight, and the careful operational procedures that characterize the industry.
Yet this same characteristic reflects something profound: nuclear power harnesses the same energy that powers stars. The fission products that create decay heat are the same elements forged in supernova explosions billions of years ago. When we split uranium, we tap into energies that shaped the universe.
Understanding this physics transforms the question from “why can’t we turn it off?” to “how do we control something so powerful?” The answer—through multiple barriers, inherent safety physics, redundant systems, and constant vigilance—represents some of humanity’s most sophisticated engineering. The reactors operating today are testaments to our ability to harness fundamental forces of nature for practical purposes, with all the complexity and responsibility that entails.
References
-
World Nuclear Association. (2025). “Physics of Uranium and Nuclear Energy.” https://world-nuclear.org/information-library/nuclear-fuel-cycle/introduction/physics-of-nuclear-energy
-
World Nuclear Association. (2025). “Nuclear Power Reactors.” https://world-nuclear.org/information-library/nuclear-power-reactors/overview/nuclear-power-reactors
-
U.S. Energy Information Administration. (2025). “Nuclear Explained.” https://www.eia.gov/energyexplained/nuclear/
-
Touran, N. (2023). “What is afterglow/decay heat?” What Is Nuclear. https://whatisnuclear.com/decay-heat.html
-
International Atomic Energy Agency. (2025). “What is Nuclear Energy?” https://www.iaea.org/newscenter/news/what-is-nuclear-energy-the-science-of-nuclear-power
-
OECD Nuclear Energy Agency. (2025). “Chernobyl: Chapter I. The Site and Accident Sequence.” https://www.oecd-nea.org/jcms/pl_28271/chernobyl-chapter-i-the-site-and-accident-sequence
-
Wikipedia. (2025). “Fukushima Nuclear Accident.” https://en.wikipedia.org/wiki/Fukushima_nuclear_accident
-
Wikipedia. (2025). “Chicago Pile-1.” https://en.wikipedia.org/wiki/Chicago_Pile-1
-
U.S. Department of Energy. (2025). “Nuclear 101: How Does a Nuclear Reactor Work?” https://www.energy.gov/ne/articles/nuclear-101-how-does-nuclear-reactor-work
-
Nuclear Power. (2025). “Six-Factor Formula - Effective Multiplication Factor.” https://www.nuclear-power.com/nuclear-power/reactor-physics/nuclear-fission-chain-reaction/six-factor-formula-effective-multiplication-factor/
-
Canadian Nuclear Safety Commission. (2025). “Nuclear Power Plant Safety Systems.” https://www.cnsc-ccsn.gc.ca/eng/reactors/power-plants/nuclear-power-plant-safety-systems/
-
Wikipedia. (2025). “Nuclear Reactor Safety System.” https://en.wikipedia.org/wiki/Nuclear_reactor_safety_system
-
World Nuclear Association. (2025). “Small Modular Reactors.” https://world-nuclear.org/information-library/nuclear-power-reactors/small-modular-reactors/small-modular-reactors
-
IAEA. (2025). “Six Global Trends in Nuclear Power You Should Know.” https://www.iaea.org/newscenter/news/six-global-trends-in-nuclear-power-you-should-know
-
Visual Capitalist. (2025). “Ranked: Nuclear Power Capacity by Country.” https://www.visualcapitalist.com/ranked-nuclear-power-capacity-by-country-2025/
